Introduction
Live cell assays constructed with genetically encoded, fluorescent biosensors can provide significant advantages over end-point assays measured in cell lysates because functional information about the timing and location of cellular responses can be monitored in cells that are relevant to disease. Fluorescent biosensors have been tools in basic research and have now been optimized for detection on automated plate readers.1 We previously demonstrated that Montana Molecular’s live cell cADDIS assay for cAMP on the CLARIOstar produces high Z’ values characteristic of a robust screening assay.2 Here we show how second messengers relevant to the Gq signalling pathway can be detected in living cells using assays for DAG, PIP2, and Ca2+. By combining spectrally distinct sensors in a single assay, we show that two responses can be detected simultaneously. Sensitive detection of these responses is enabled by employing the CLARIOstar microplate reader using filters or the LVF monochromator.
Assay Principle
Montana Molecular’s biosensors achieve optimal expression in mammalian cells due to packaging in a modified baculovirus (BacMam). Figure 1 shows the schematic of the mechanism for the DAG sensor, in which a circularly permuted fluorescent protein is inserted near the DAG binding domain of protein kinase C. Similar sensors are used for detecting Ca2+ and PIP2.
Each sensor comes in either a red or green version which can be spectrally resolved. By combining red and green sensors in a single transduction step, changes in two-second messengers can be detected simultaneously. The DAG biosensors are available as decreasing in fluorescence (Downward DAG) or increasing in fluorescence (Upward DAG). Both versions indicate increases in diacylglycerol in living cells.
Gq Detection using DAG, PIP2, and Ca2+ sensors
Montana Molecular has developed a suite of biosensors for the unambiguous detection Gq signalling.3,4 Figure 2 illustrates hM1R mediated Gq signalling in response to Carbachol. We validated sensors for PIP2, Diacylglycerol (DAG) and Ca2+ on the CLARIOstar microplate reader.
Materials & Methods
- HEK293 cells
- Montana Molecular DAG sensors, PIP2 sensors, R-GECO Ca2+ sensors packaged in BacMam
- Greiner 96-well F bottom plates
- CLARIOstar microplate reader from BMG LABTECH
Transduction of the indicated sensor carried in BacMam was performed on HEK293 cells in suspension. Cells were subsequently plated in 96-well microplates and sensor expression allowed to proceed for 24-36 hours. Thirty minutes prior to the experiment media was exchanged for PBS and cells were allowed to rest at room temperature.
Instrument Settings
| Detection Mode: | FI (well mode), bottom optic |
| Number of flashes: | 5 |
| Scan mode: | Orbital |
| Scan diameter (mm): | 2 |
| Gain / Focal height: | Adjusted prior to test run |
| Interval time: | 3-11 s |
| Optical Settings | Green | Red |
| Excitation: | F 482-16 | 560-15 |
| Dichroic: | LP 504 | Auto580.5 |
| Emission: | F 530-40 | 618-49 |
Injection Settings
| Volume (μl): | 50 |
| Pump speed (μl/s): | 85 |
| Injection start time (s): | 30 |
Results & Discussion
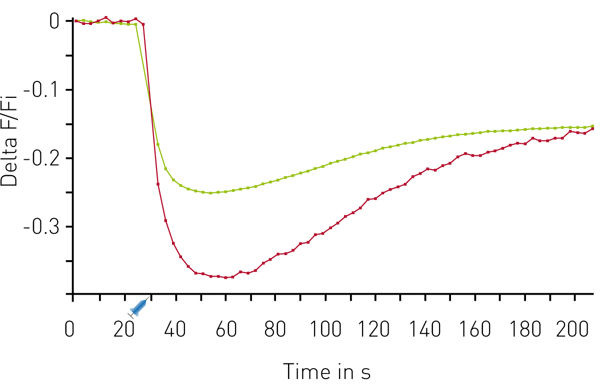
The results in figure 3 show that the DAG sensor exhibits excellent assay performance.
Figure 4 shows the results of a response to carbachol in a multiplexed experiment. The results show the very rapid calcium release as well as rapid production of DAG following compound injection.
Figure 5 shows another multiplexed experiment. In this case, using the Downward DAG sensor and a sensor for PIP2.
Conclusion
The CLARIOstar supplies high sensitivity detection of the genetically encoded Montana Molecular sensors. Spectrally-resolved variants indicate simultaneous changes in multiple second messengers following Gq activation.
References
- Tewson, P., et al (2016) J. Biomol. Screen. 21:298-305
- Tewson, P., et al (2016) Real-Time Detection of Gs and Gi Signalling in Living Cells
- Ding, Y., et al (2015) Nature Methods 12:195-198
- Tewson, P., et al (2012) Simultaneous Detection of Ca2+ and Diacylglycerol Signaling in Living Cells




![Fig. 3: DAG assay performance. Comparison of 14 replicates treated with Carbachol (circle) or vehicle [PBS] (purple circle) indicates robust assay performance. Z’ = 0.8.](https://www.bmglabtech.com/hs-fs/hubfs/1_Webseite/5_Resources/App%20Notes/5_App%20Notes%20300-349/AN301-Fig3.webp?width=600&height=526&name=AN301-Fig3.webp)