
NEPHELOstar Plus
Microplate nephelometer for light-scattering and turbidity measurements


Explore nephelometry for precise turbidity measurements in solution-based assays. Learn how microplate readers enhance sensitivity for applications in drug formulation, protein aggregation, and particle analysis.
Nephelometry is an analytical chemistry technique that measures the intensity of light scattered by insoluble particles in a solution. The word itself comes from the Greek nephelo, meaning cloud, reflecting how suspended particles create a cloudy or turbid appearance. By detecting this scattered light, nephelometry quantifies the level of turbidity in a sample.
When directed through a turbid solution containing suspended solid particles, light is transmitted, absorbed (blocked), and scattered (reflected off the particles; fig.1). The amount of scattered light depends on the size, shape, and concentration of the insoluble particles in the solution, as well as on the incident wavelength of light.
Theories and concepts on light scattering were initially undertaken at the end of the 19th and beginning of the 20th century, mainly by Rayleigh, Mie, and Debye.
Nephelometry was first applied in the field of clinical chemistry to immunoassays for the detection and quantification of serum proteins in the blood, such as immunoglobulins and macromolecules. These applications are still in use today. Applied to microplate readers, they are mainly used to analyze material precipitation (fig. 2), such as drug solubility and protein aggregation, or bacterial growth.
Light scattering in liquids follows the rules of elastic scattering of particle physics, in which no energy is absorbed by either particle during the “collision”. The energy of a photon before and after the scattering event is not changed. Elastic scattering is different in large and small particles. For large particles, light is scattered mainly in the forward direction (forward-angled). When the size of the particles is smaller than 5% of the wavelength of light by which they are hit, scattering is symmetrically distributed.4
Soluble molecules are generally small in size (compared to the wavelengths of incident light) and scatter in an almost symmetrical way (fig. 4A). In contrast, precipitates and complexes in solution have typically a larger size (closer to the wavelengths of incident light), predominantly producing a forward-angled scatter (fig.4B). Nephelometric detection typically focuses on measuring forward scatter.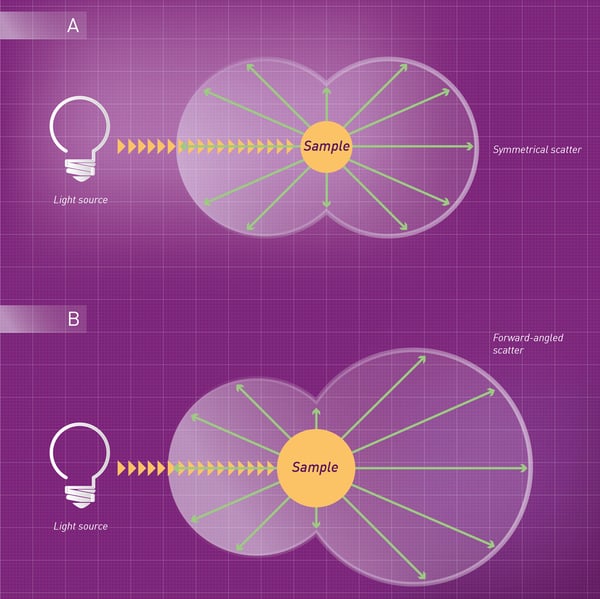
The intensity of scattered light (IS) and the precipitate´s concentration (C) are related by the following equation:
IS = kS * I0 * C
where kS is a constant determined from a calibration of the system and I0 is the intensity at the light source.
The physical properties of a suspension of particles are influenced by different variables. Although scattering is related to the concentration of solid particles in solution, the intensity of the scattered light also depends on their size and shape. Equally concentrated samples containing precipitates of differing sizes will show different scattering levels.
Additionally, precipitate size and shape are affected by temperature, pH, and reagent concentration, as well as the order of mixing, stirring, and the interval between the formation of precipitate and detection. To obtain reproducible conditions and results among samples and assays, it is necessary to consider all these different variables. 4, 5
Wavelength selection is usually irrelevant as the absorption of the incident light by the particles in suspension is generally not considered, provided it does not induce fluorescence of the sample. Thus, if non-fluorescent samples are used, there is no specific need for wavelength selection. The choice of the wavelength is based primarily on the need to minimize potential interferences and rather affects the intensity of the incident light or of the scattering itself.4
Turbidity or cloudiness can be detected by either nephelometry or turbidimetry. Both techniques are non-destructive and based on light scattering caused by suspended solid particles, but they are not the same - despite sometimes being used interchangeably.
Turbidimetry measures the loss of light intensity caused by the scattering effect of insoluble particles as it passes through a sample. A detector placed in line with the light source captures the transmitted light, and the decrease in transmission is compared to a reference and expressed as Optical Density (OD) units. This means turbidimetry can be performed using a standard absorbance microplate reader.
Nephelometry, by contrast, directly measures the intensity of light scattered by particles in the sample. The detector is positioned at an angle to the light source to avoid interference from transmitted light.
The choice between nephelometry and turbidimetry is primarily guided by two factors:
Accordingly, nephelometry is best suited and offers higher sensitivity for the analysis of small, suspended particles at low concentrations, whereas turbidimetry is usually applied to relatively large insoluble particles at high concentration such as measuring cell density in bacterial cultures.3
Although fluorometers can be employed for nephelometric detection, the angular dependence of scattering encouraged the development of dedicated devices. Turbidimeters with detectors located at an angle to the incident beam are called nephelometers and are considered the standard instrument for the measurement of low turbidity values. They measure the intensity of scattered light. Transmitted light is not detected.
The basic components of this device include a light source, light-scattering optics, and a detector. The light source generates a beam that is directed through the sample. Halogen and xenon lamps or lasers can be used as light sources. Lasers are typically the most common choice, due to their sensitivity, high intensity, and coherent nature (emitted photons are “in step” with each other). Incoming and outcoming wavelengths are identical and hence not optically selected.
A detector is placed opposite the light source and at an angle relative to the incoming light beam. It detects variations in either forward-angled scatter or side scatter, depending on its position. Depending on the angle at which the most scattering can be collected, detectors may be placed at angles of 30°, 70°, or 90°.
Nephelometry can be conducted as both, endpoint or kinetic measurement. Endpoint measurements quantify the maximum light scattering after a reaction reaches equilibrium or at a pre-determined time point. Kinetic detection (multiple readings over time) can be applied throughout the whole precipitation process and usually provides more information on the reaction.
NTU (Nephelometric Turbidity Units) is the standard unit for expressing turbidity measured by a nephelometer. Instruments are calibrated using formazin, a polymer suspension that produces highly reproducible light scattering, providing a consistent reference point across devices. NTU is widely used in water quality monitoring to assess the clarity of drinking water, wastewater, and environmental samples.
Since the 1970s immunonephelometry has been applied in clinical laboratories for the analysis of immunoassays. It was originally used to detect the formation and precipitation of immune complexes (antigen-antibody), an application that is still in use today. Immunonephelometry is also used to determine the concentration of serum proteins, including immunoglobulin, as well as in high-volume automated coagulometers. These devices quantify coagulation factors in blood samples and allow for multiple-assay coagulation profiles.
In pharmaceutical laboratories, nephelometry is mainly used to assess the solubility of drugs or compounds. Furthermore, it is a promising method for the quantification of microbial growth and is commonly used to determine cell counts of microorganism suspensions such as yeast (e.g. S. cerevisiae). 6
Nephelometry can also be detected in microplates (fig.5). This format is especially advantageous for life science laboratories and the pharmaceutical industry since microplate-based handling of samples and compounds increases both efficiency and throughput. Microplate-based nephelometers usually provide higher throughput, simplified, and low-volume approach to the collection of turbidity data.The world’s first laser-based nephelometric detection device in microplates, the NEPHELOstar Plus, was developed by BMG LABTECH. This instrument detects particles in solution in a microplate well by measuring the forward-angled scattered light generated when a laser beam is directed through the sample.
Microplate-based nephelometry is an invaluable tool for the pharmaceutical industry due to its applicability in high throughput compound solubility screenings. Additionally, it can be used for microbial growth and protein binding kinetics, to measure calcification propensity in body fluids, rheumatoid factors in serum, antigen-antibody binding (fig. 9), and much more.
In the pharmaceutical industry, high-throughput screening is an important method for drug discovery. The assessment of solubility in this process is mandatory to determine the validity of the pharmacological results and the selection of promising compounds. Drug Solubility has a major impact on drug availability, formulation, dosing, and absorption. Hence, it is very important to analyze it early in the drug discovery process to avoid time-consuming and costly ADME screens of low solubility compounds.
Traditionally, equilibrium solubility assays have been determined in limited throughput, by shaking and incubating the compound with a solvent for at least 24h, prior to filtration and concentration determination by HPLC. This approach no longer fulfills the requirements of modern drug discovery.
Today, automated kinetic solubility screens run on microplate nephelometers deliver higher throughput in shorter times. In this approach, a serial dilution of the compound to be tested is prepared in an aqueous solution and pipetted on a microplate. The undissolved precipitate is detected by light scattering. At high concentrations, the compound will precipitate making the suspension cloudy and delivering high RNUs counts. As long as the concentration is higher than the solubility, the compound will precipitate. When the concentration is lower than the solubility, the compound will completely dissolve resulting in a clear solution. Accordingly, the scatter and the RNU counts will be significantly reduced. Typically, two linear fittings for the soluble and insoluble phases are applied to the data. The point at which they cross is taken as the kinetic solubility point (fig.10).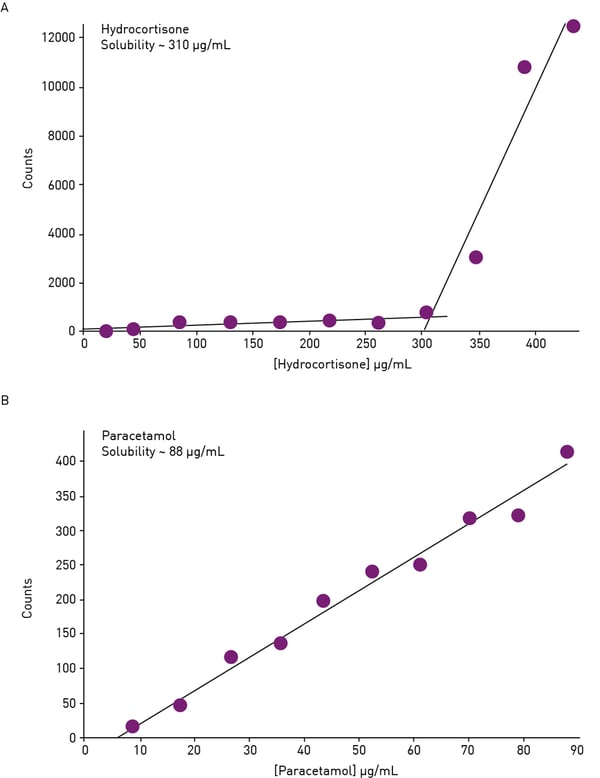
Advantages of this approach are the speed of the assay and the ease of handling. Microplate-based nephelometric assays require only pipetting steps; filtration or phase separation of the solution from the undissolved residue is not needed. Moreover, no liquid transfer step is required as the setup of the assay and its measurement can be performed in the same microplate. Finally, it can be employed to determine both the concentration at which a compound becomes soluble and the point at which a solute begins to precipitate.
Typically, the detected signal is linear for up to 3 orders of magnitude of particle concentration and a limit of detection of about 20 mmol/L can be reached for kinetic solubility assays.8
Microplate-based nephelometry can also be used as an alternative to absorbance-based OD600 microbial growth detection. As bacteria multiply, the number of cells fluctuating in solution will increase, thus increasing scattering levels and RNU counts. A serial dilution of the culture is generally used to relate optical density to RNU counts. The nephelometric approach is comparable to the absorbance-based one but usually bears a higher sensitivity.
The application notes Monitoring of microbial growth curves by laser nephelometry and Nephelometric monitoring growth of Candida albicans (fig.11) show how growth curves can be efficiently measured on the NEPHELOstar Plus.
In research laboratories, nephelometry is a versatile technique with many time-saving applications. In the research laboratory, nephelometry is often used to study immune responses, investigate immune mechanisms, or look at immune events. Many applications centre around protein quantification.
Microplate nephelometer for light-scattering and turbidity measurements