Introduction
In order to understand protein structure, dynamics and energetics it is clear that one must understand the kinetics of protein folding that underlie protein stability. It is desirable to perform φ-value analyses, however collecting data for these analyses is laborious and in most cases, the methods are not amenable to high throughput.
Here we describe the adaptation of previous methods using the ability of the Omega microplate reader to measure tryptophan (Trp) fluorescence. The method also takes advantage of the reagent injection option to develop a technique that allows for direct measurement of the unfolding rates of proteins.
Assay Principle
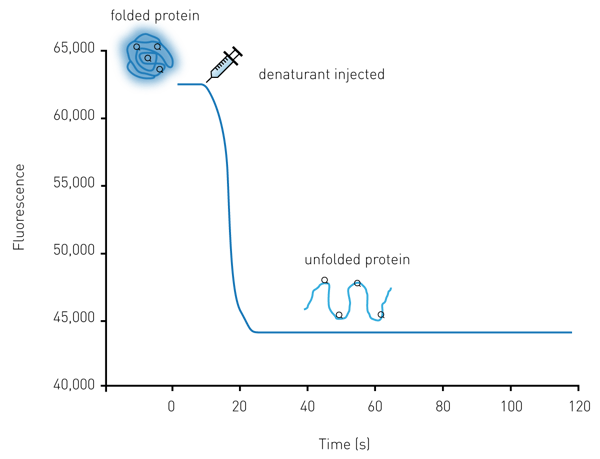
Two assay approaches were developed. The kinetic protein unfolding assay (Figure 1) measures protein unfolding over time by injecting the denaturant into a well and monitoring fluorescence emission of the intrinsic tryptophan residues. The injection at the point of measurement feature of the Omega allows for the retention of the maximum amount of signal information.
 For proteins with an unfolding kinetic constant of greater than 0.01s-1 a limited proteolysis assay was developed. Varying concentrations of either trypsin or thermolysin were manually added to the protein and monitoring of Trp fluorescence was performed over time.
For proteins with an unfolding kinetic constant of greater than 0.01s-1 a limited proteolysis assay was developed. Varying concentrations of either trypsin or thermolysin were manually added to the protein and monitoring of Trp fluorescence was performed over time.
Materials & Methods
-
96-well Quartz microplates (Hellma)
-
POLARstar Omega microplate reader (BMG LABTECH)
For detailed experimental procedures please refer to Wang et al.1. Proteins tested included AbpSH3 (the src homology 3 domain from Actin Binding Protein 1), and mutants of this protein: AbpSH3 E7L, AbpSH3 V21K, and AbpSH3 Triple which contains the mutations E7L, V21K, and N23G. Next to that an AbpSH3 – ArkA (Actin Regulating Kinase SH3 binding peptide A) hybrid named HLL (hybrid long linker) was tested.
Omega Instrument Settings
| Kinetic protein unfolding assay |
|
| Measurement type: |
FI (Bottom) |
| Measurement mode: | Well Mode |
| No. of cycles: |
1000 |
| Cycle time: |
120 ms |
| Filters: | Ex: 280-10 / Em 330-10 |
| Gain: | 1400 |
| Injection volume: |
4.0 – 92.0 μl (4.0 μl increments) |
| Limited proteolysis unfolding assay |
|
| Measurement type: | FI (Bottom) |
| Measurement mode: | Well Mode |
| No. of cycles: |
1000 |
| Cycle time: |
73 s |
| Filters: | Ex: 280-10 / Em 330-10 |
| Gain: | 1400 |
Results & Discussion
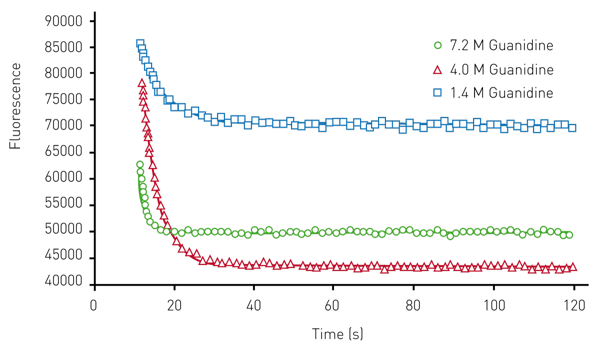
Representative data that was achieved for the wild-type AbpSH3 protein using the Omega plate reader is shown in (Figure 2). These data are the unfolding curves generated at different concentrations of the denaturant guanidine.
From these data, Chevron plots of the unfolding can be determined (data not shown) which allow for the determination of unfolding constants shown in Table 1. Note that the values determined for AbpSH3, as well as mutant forms of this protein, are close to those previously reported in the literature2,3. This indicates that this assay is comparable to stopped-flow approaches.
Table 1: Summary of kinetic unfolding data
| ku exp. (s-1) X 103 |
ku literature (s-1) X 103 | mu exp. (kJ mol-1 M-1) | mu literature (kJ mol-1 M-1) | |
| AbpSH3 WT | 71.0 ± 5.4 | 65.8 ± 5.3 | 0.92 ± 0.06 |
0.83 ± 0.04 |
| AbpSH3 E7L | 7.2 ± 0.5 | 4.8 ± 1.6 | 1.09 ± 0.01 |
0.96 ± 0.10 |
| AbpSH3 V21K | 48.5 ± 10.5 | 44.8 ± 4.3* | 0.69 ± 0.05 |
0.56 ± 0.03* |
| AbpSH3 Tri. | 2.9 ± 0.6 | 4.7 ± 3.6 | 1.01 ± 0.01 |
0.70 ± 0.20 |
| HLL | 4.2 ± 0.8 | n/a | 1.07 ± 0.01 |
n/a |
*Experiments used urea instead of guanidine as denaturant
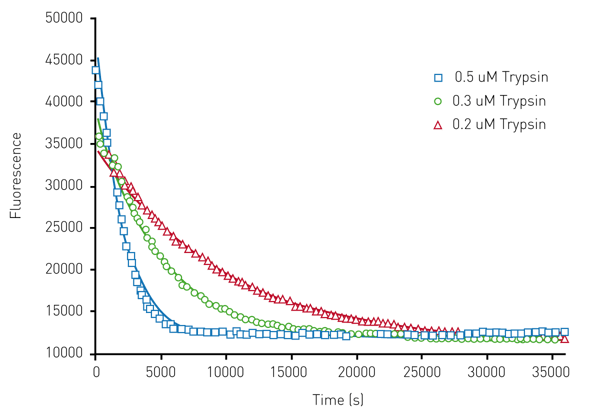
Figure 3 shows the results of the limited proteolysis unfolding assay using HLL which show unfolding monitored in the presence of trypsin.
A Michaelis-Menten type equation was used with this data, yielding unfolding constants of 0.0023s-1 for trypsin. Note that a value of 0.0042s-1 is shown in Table 1. From these results, we conclude that this complementary method is a viable alternative for fast unfolding proteins. Thus, both the chemical denaturation and limited proteolysis approaches, provide reliable high throughput kinetic protein unfolding measurements on the Omega.
Conclusion
The kinetic protein unfolding assay has several advantages over previously described methods. First, it has a shorter dead-time for the assay meaning proteins that unfold relatively quickly can be monitored. The ability of the Omega to take readings every 120 ms in this assay is of further benefit. Both of the assays described here can be performed in a relatively low volume (100 µl) at protein concentrations that will conserve samples.
References
- 1. Wang Q. et al. (2016) Development and Application of a High Throughput Protein Unfolding Kinetic Assay PLoS ONE 11 (1):e0146232.
- Rath A and Davidson AR (2000) The design of a hyperstable mutant of the Abp1p SH3 domain by sequence alignment analysis Protein Sci. 9(12):2457- 2469.
- Maxwell KL et al. (2005) Protein folding: defining a “standard” set of experimental conditions and a preliminary kinetic data set of two state proteins Protein Sci. 14(3):602-616.