Introduction
Epigenetics is an emerging arena of science in which changes in the regulation of gene activity and expression are not dependent on the sequence of genes. Instead, the changes in expression are the result of DNA methylation or modification of histones. These modifications are essential to the normal developmental process and the results can be devastating when the epigenetic process is disrupted leading to cancer and autoimmune disease. The realization of the importance of epigenetic regulation has led to the search for drugs which are capable of targeting the enzymes that add or remove epigenetic modifications. In order to perform screening to identify compounds which affect epigenetic modifying enzymes, it is necessary to establish a reliable assay which is capable of monitoring the activity of these enzymes. Cisbio has made available a number of assays that have been verified to detect the activity of the enzymes involved in histone modifications. These enzymes fall into two broad classes:
- Writers: enzymes that add methyl or acetyl groups
- Erasers: enzymes that remove methyl or acetyl group
Here we describe the verification of the performance of a Cisbio HTRF® based assay using the PHERAstar FS from BMG LABTECH.
Assay Principle
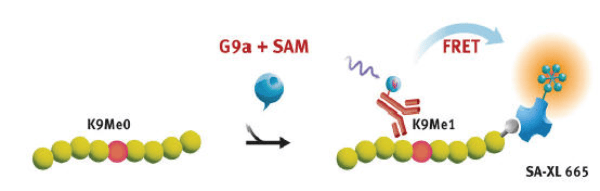
The assay is a Histone H3K9 monomethylation assay that uses a biotinylated peptide that comprises residues 1-21 of histone H3 in which lysine 9 is un-methylated as a substrate (Figure 1).
The assay is performed without washing in a single well using an enzymatic step followed by a detection step. In the enzymatic step substrate, enzyme G9a, and S-(5’-Adenosyl) L-methionine chloride (SAM) are combined. During incubation, active enzyme will transfer a methyl group to the lysine residue resulting in methylated substrate. The amount of modified substrate is detected via addition of a Eu3+-cryptate labeled anti- H3K9 me1 detection antibody and XL665-conjugated streptavidin. The antibody will bind specifically to the methylated substrate and fluorescence resonance energy transfer will be observed between HTRF donor (antibody) and acceptor (XL665).
Materials & Methods
- H3K9 mono-methylation detection reagents and protocol (Cisbio)
- 384 well, small volume, white microplates (Greiner)
- G9a (Reaction Biology Corp.)
- H3K9 peptide substrate (AnaSpec)
- SAM & SAH (Sigma)
- UNC0646 & BIX01294 (R&D Systems)
- PHERAstar FS (BMG LABTECH)
- 4 μl inhibitor or enzymatic buffer
- 2 μl G9a (incubate for 5 minutes at room temperature)
- 4 μl substrate/SAM mix (seal plate and incubate at room temperature)
- 10 μl of detection mixture
Data analysis
The HTRF ratio (Signal at 665nm / Signal at 620nm; the result is multiplied with 10,000) is automatically calculated by the MARS Data Analysis Software.
Results & Discussion
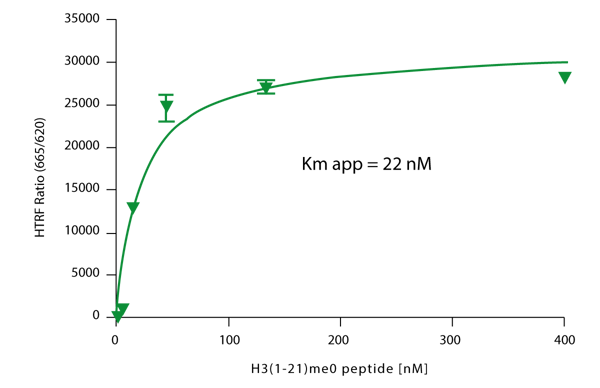
Previous results indicated optimal performance by using a G9a concentration from 0.125-0.25 nM in 40 to 60 minute reaction time. Therefore the experiments described here employ a G9a concentration of 0.12 nM and a 40 minute reaction time. To verify performance a substrate titration was performed to determine the Km for the peptide in this experimental protocol. The results of this titration experiment are shown in Figure 2.
 Final validation of the G9a H3K9 methylation assay was obtained by measuring the activity of known G9a inhibitors (Figure 3).
Final validation of the G9a H3K9 methylation assay was obtained by measuring the activity of known G9a inhibitors (Figure 3).
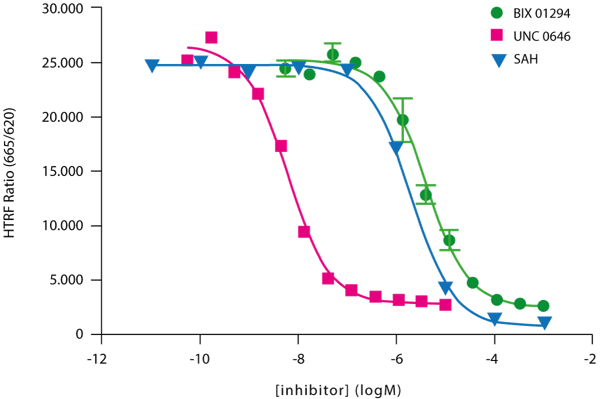 IC50 values were calculated from enzyme inhibition curves similar to those shown in Figure 3. The IC50 results, are presented in Table 1. Table 2 compares the read times of the PHERAstar FS when flash number is varied.
IC50 values were calculated from enzyme inhibition curves similar to those shown in Figure 3. The IC50 results, are presented in Table 1. Table 2 compares the read times of the PHERAstar FS when flash number is varied.
Table 1: IC50 values for three different G9a inhibitors
| Inhibitor | IC50 |
| BIX 01294 | 4.1 μM |
| UNC 0646 | 6.2 nM |
| SAH | 1.9 μM |
Table 2: Read times for varying TRF laser conditions in a 384 well plate.
| Laser mode: | Precise (27 flashes) | Rapid (7 flashes) | Flying (1 flash) |
| Read time: | 2m 32sec | 48 sec | 21 sec |
Conclusion
- The Cisbio HTRF® G9a Histone H3K9 monoMethylation Assay provides an excellent platform to screen for inhibitors of these enzymes with a central role in epigenetic regulation
- Assay performance on the PHERAstar FS is verified using substrate, cofactor and inhibitor titrations
- One flash fly mode significantly improves read times which will increase throughput
- Excellent performance in one flash fly mode can be attributed to:
- UV laser as an excellent excitation source
- HTRF® specific modules which provide true simultaneous dual emission detection
- Matched PMT’s designated for TRF detection