Introduction
The quantification of nucleic acids samples is a major need in any genetic and molecular biology laboratory. Next to well-known UV absorbance assays (e.g. 260/280 nm), a lot of different fluorescence intensity based DNA quantification assays are offered. These assays are not only more sensitive than absorbance assays, they are also able to differentiate between double-stranded and single-stranded DNA, as well as RNA.
Aside from sensitivity and specificity, the demand for very low volume measurements is emerging. This is challenging for assays as well as for the detection instrumentation. In this application note, we present DNA quantification data obtained on the low volume LVis Plate, as well as 96- and 384-well microplates.
Materials & Methods
- Quant-iT™ PicoGreen® dsDNA assay, LifeTechnologies
- Low volume LVis Plate, BMG LABTECH
- Black 96-well and 384-well microplates, Greiner
Sample preparation
All necessary reagents such as detection reagent, TE buffer, DNA standard (Lambda DNA standard) are provided with the kit. From a 20x TE buffer stock solution, a 1x TE buffer was prepared using HPLC grade water. The PicoGreen® reagent was diluted 1:200 by using 1x TE buffer. dsDNA standards were diluted with 1x TE buffer to obtain a concentration range of 2 to 1000 ng/ml. DNA standards were pipetted in 6 replicates, adding the same amount of PicoGreen® reagent into each well leading to the final volumes: 200 µl in 96-well plates, 20 µl in 384-well plates, and 2 µl in the LVis Plate. The blank consisted of TE buffer.
Instrument settings
|
FLUOstar Omega
|
CLARIOstar
|
PHERAstar FS
|
|
|
Filter settings
|
Ex485
Em520
|
Ex485
Em520 |
Optic Module:
FI 485 520
|
|
Monochromator settings
|
Ex: 483-14
Em: 530-30 |
All other settings are default settings for a fluorescence endpoint test protocol.
Results & Discussion
Fluorescence scan of PicoGreen® reagent bound to DNA
In order to determine useful monochromator settings for the CLARIOstar, an excitation and emission scan was done for the DNA bound PicoGreen® reagent. The spectra scan result is shown in Fig. 1.
The scan showed an excitation maximum at 502 nm and an emission maximum at 524 nm. For further measurements, the highest signal/blank ratio was determined by varying the band widths for excitation and emission.
96-well
In 96-well plates the standard curves obtained using either filters or the monochromator are very similar (Fig. 2). With filters, the slope of the standard curve is slightly higher.
384-well
To miniaturize the assay, the well volume was decreased to 20 µl. The resulting standard curve in 384-well small volume plate (Fig. 3) was comparable to the data obtained for 96-well plates.
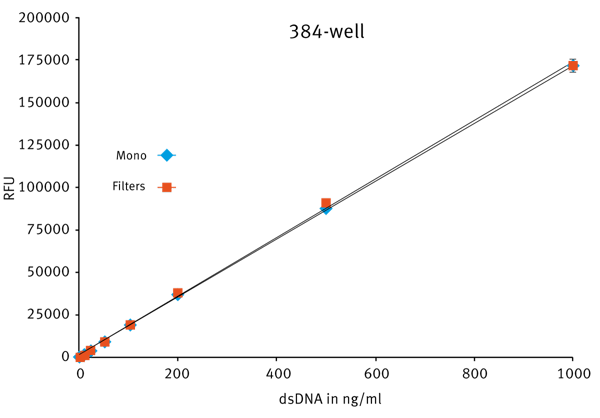 Very good linearity was achieved for the CLARIOstar using either filters (R2 = 0.9999) or LVF Mono-chromators™ (R2 = 0.9998).
Very good linearity was achieved for the CLARIOstar using either filters (R2 = 0.9999) or LVF Mono-chromators™ (R2 = 0.9998).
LVis Plate results
With the LVis Plate, it is possible to miniaturize the assay further. This special plate was developed to use a volume of as little as 2 µl.
Originally developed to do DNA and protein UV absorbance quantification the LVis Plate, used in the CLARIOstar or PHERAstar FS, is an excellent tool to determine the DNA content of samples in fluorescence intensity mode. Results for the PicoGreen® assay in the LVis Plate are shown in Fig. 4.
Conclusion
The standard curve data show that the PicoGreen® assay can be measured with high linearity and sensitivity over a broad DNA concentration range. For the CLARIOstar, the LVF Monochromator™ shows filter-like performance. For example, the sensitivity in 384-well plates was calculated to be about 3 pg/well for both filters and LVF Monochromators™.
With the help of the PicoGreen® assay, any preferred volume, between 2 µl in the LVis Plate or standard volumes in 96- and 384-well plates, can be used to determine the DNA concentration of samples.
PicoGreen is a registered trademark of Invitrogen.




