Introduction
Determining total protein concentration is an accepted means of comparing and standardizing biological samples. The utility of protein concentration assessment extends to high-throughput screening platforms including proteomic and genomic applications. Ideally the protein concentration determination should also be amenable to high throughput. Most protein determination assays use absorbance measurement detection which is difficult to minimize for higher throughput. Previously, it has been reported that colorimetric assays, including the bicinchoninic acid (BCA) protein assay, can be performed in white plates using fluorescence detection1,2. The method exploits the inherent fluorescence of white plates. In presence of an absorbing solution, the inherent fluorescence is quenched and the decrease in fluorescent signal can be used to measure colorimetric assays.
Assay Principle
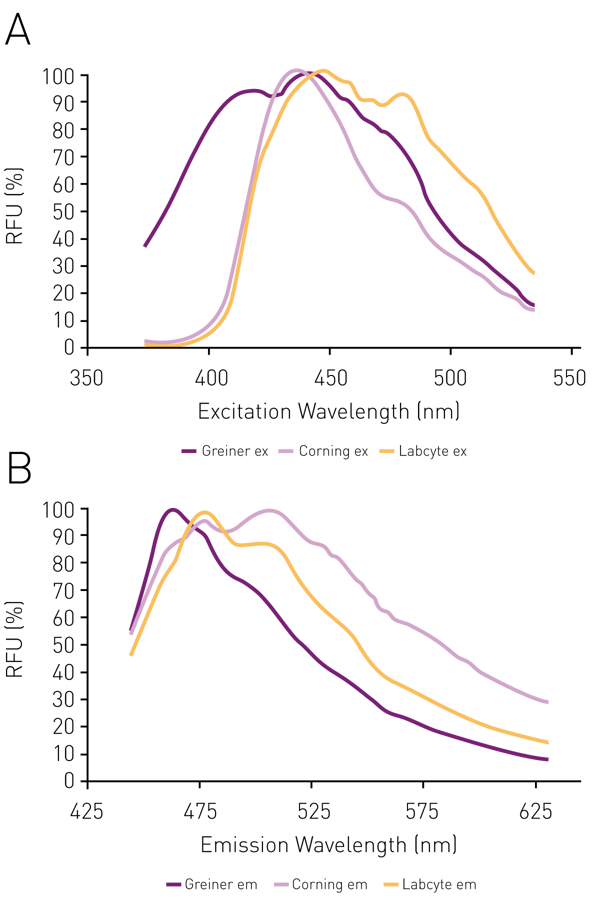
The epi-absorbance approach described here relies on the fact that white plates exhibit fluorescent characteristics such that when an excitation light of ~ 435 nm is shined on the plate an emission signal at about ~ 560 nm can be observed (Figure 1). The colorimetric reagent/product absorbs light and suppresses the fluorescent signal that can be detected. Further development of the colorimetric product leads to a further decrease in fluorescent signal detected.
Materials & Methods
- PierceTM BCA Protein Assay Kit and Pre-Diluted Protein Standards: Bovine Serum Albumin (BSA) Set (Thermo Scientific)
- White, low volume 384 well plates (Greiner/Corning)
- White, 1536 well plates (Labcyte)
- CLARIOstar® microplate reader (BMG LABTECH)
Spectral scanning
Empty wells and wells containing prepared BCA reagent with or without varying concentrations of BSA were added to each plate type. These were scanned with the CLARIOstar using the following settings:
| Excitation Scan | Emission Scan | |
| Excitation wavelength [nm] | 374 -> 534 | 417 |
| Excitation bandwidth [nm] | 10 | 16 |
| Emission wavelength [nm] | 562 | 445 -> 630 |
| Emission bandwidth [nm] | 16 | 10 |
| Gain (Greiner 384) | 1599 | 1254 |
| Gain (Corning 384) | 1506 | 1416 |
| Gain (Labcyte 1536) | 1290 | 996 |
Fluorescent detection of BCA Assay
Replicates of each BSA concentration were added to the plates. A volume of 2 μl was used for 384 well plates and a volume of 1.5 μl was used for 1536 well plates. Prepared BCA reagent was subsequently added. A volume of 10 μl was used in 384 well plates and 7.5 μl in 1536 well plates. The plates were read on the CLARIOstar with the settings indicated below.
| Excitation | 435 - 15 |
| Dichroic | 497.2 |
| Emission | 562 - 20 |
| Gain (Corning) | 1758 |
| Gain (Greiner) | 1729 |
| Gain (Labcyte) | 1407 |
Data were transformed to enable analysis with a linear regression fit.
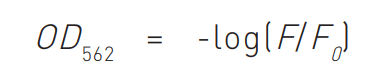 F= fluorescence of sample, F0 = fluorescence of buffer blank
F= fluorescence of sample, F0 = fluorescence of buffer blank
Results & Discussion
The comparison of two low volume 384 well plates (Greiner and Corning) and a 1536 well plate (Labcyte) shows reasonably similar spectra for each plate with excitation maxima in the range of 435-450 (Fig 2 A) and emission maxima in the range of 460-510 (Fig. 2 B).
BCA reagent decreases both, fluorescence excitation and emission peaks. A further decrease is observed in the presence of increasing protein concentrations following color development (Fig 3). 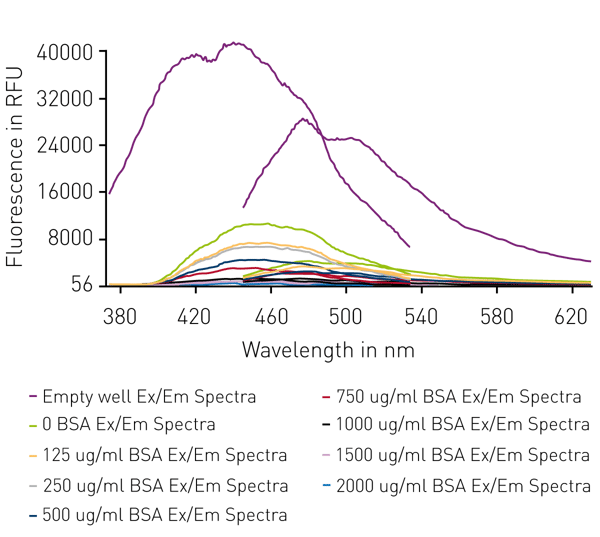
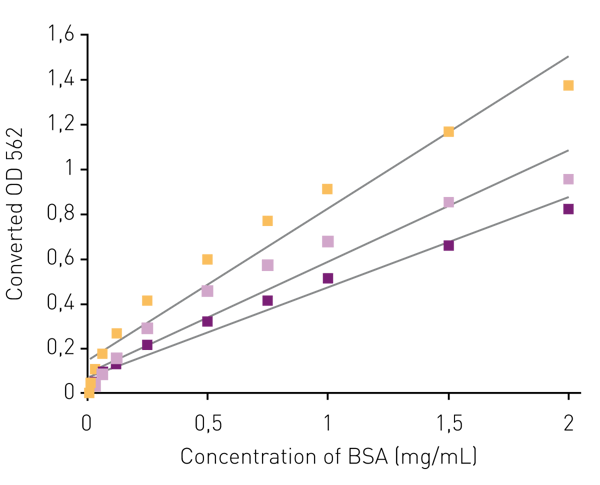
Based on this spectral data the CLARIOstar LVF monochromatorTM was set to 435-15 for excitation and 562-20 for emission and the linear variable dichroic was automatically adjusted to 497.2 to read replicates of various concentrations of BSA (125-2000 mg/mL). The fluorescence measurements were used to create transformed standard curves, which, when graphed show that the 12 μl (2 μl protein) samples in 384 well plates conform to a linear fit as do the 9 μl (1.5 μl protein) samples in 1536 well plates (Fig. 4).
Conclusion
Our results show that the epi-absorbance approach can successfully miniaturize the BCA assay. For the first time miniaturization to 1536 well plates is reported. Significant savings in reagent volume and sample volume can be achieved. Although the spectral characteristics of each plate differ slightly, these differences did not keep us from achieving direct comparisons that showed that all plates tested exhibit a good linear correlation between fluorescence data converted to OD562 and BSA concentration.
References
- Zuck PI, O’Donnell GT, Cassady J, Chase P, Hodder P, Strulovci B, Ferrer M (2005) Miniaturization of absorbance assays using the fluorescent properties of white microplates. Anal Biochem 342 254-259
- Bainor AI, Chang L, McQuade TJ, Webb B, Gestwicki JE, (2010) Bicinchoninic acid (BCA) assay in low volume Anal Biochem 410 310-312