
PHERAstar FSX
Powerful and most sensitive HTS plate reader
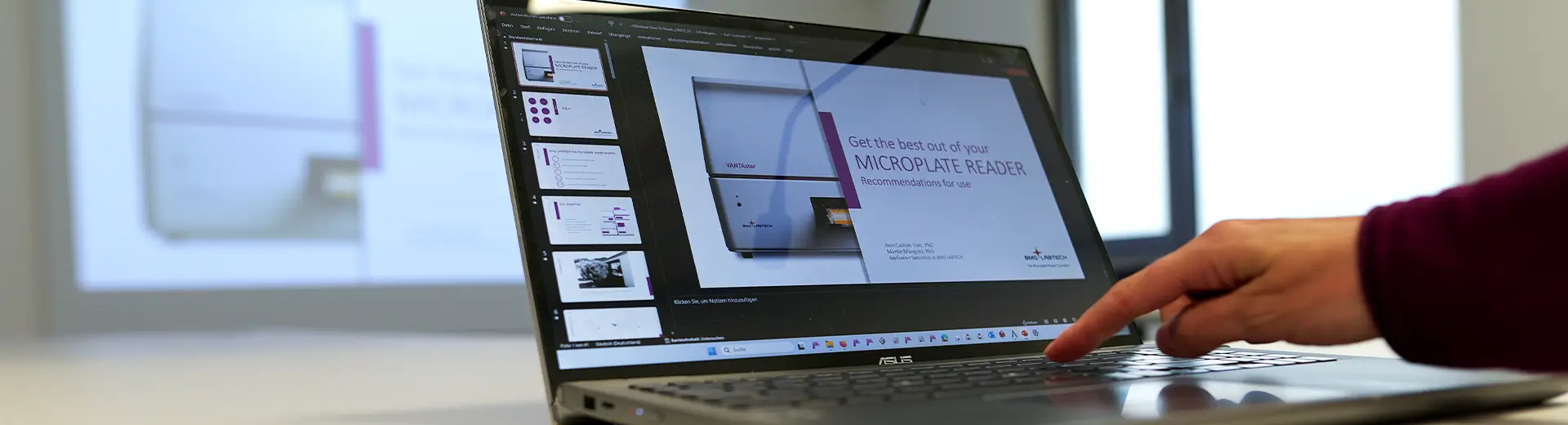
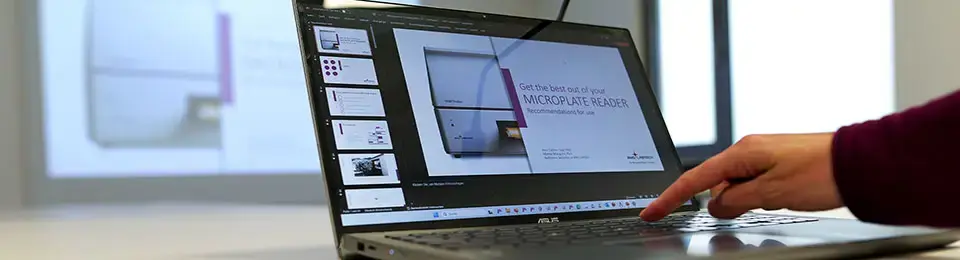
Protein kinases and phosphatases are crucial targets for drug discovery. Discover how kinetic and endpoint monitoring of their activities can facilitate target discovery and drug development.
Protein kinases are a diverse group of 518 enzymes whose dysregulation underlies 400 or more human diseases across therapeutic areas and especially oncology and inflammation. Currently, 30% of all drug development efforts are focused on protein kinases. Although many drugs are approved or in clinical trials, these are predominately ATP-competitive inhibitors. More recently, there has been a surge in the generation of kinase inhibitors with different modes of action, where new tools are needed to effectively characterize inhibitor function, predict drug potency, and drive decisions earlier in the drug development process.
PhosphoSens® Cys-Sox-based sensors harness chelation-enhanced fluorescence by integrating the sulfonamido-oxine (Sox) chromophore to create optimized peptide sensors for the quantitative and homogenous detection of kinase and phosphatase activity. The same sensor can be used for kinetic or endpoint assay formats using the CLARIOstar Plus microplate reader. The endpoint analysis employs sensitization with europium so data can be collected in a time-resolved manner at a 620 nm emission wavelength to eliminate background fluorescence, making this approach excellent for high throughput screening. The kinetic analysis allows detailed and time-dependent characterization of inhibitor mechanisms or modes of action, potency, and residence time determinations. The Sox-based assay technology can be applied across the entire drug development workflow to provide a quantum improvement in performance and productivity in addressing the opportunities of next-generation protein kinase and phosphatase inhibitors.
In this tutorial, you will learn more about these new tools, the diverse targets for which peptide substrate sensors are available, and how custom solutions can be produced to enable target discovery and drug development. Examples of using the MARS data analysis software to further improve throughput and evaluation will also be presented.

Powerful and most sensitive HTS plate reader
Most flexible Plate Reader for Assay Development
Flexible microplate reader with simplified workflows