Introduction
The development of virucidal drugs requires the screening of compound libraries to assess their impact on viral cytopathic effects and viral replication1. Plaque counting remains the gold standard for evaluating the efficacy of antiviral compounds, however, this process is very labor-intensive and time-consuming. Measuring a reduction in plaque formations is therefore only used to evaluate compounds that have already been identified as virus-specific inhibitors by other means such as their impact on the viral cytopathic effect2.
Here we describe the use of BMG LABTECH’s CLARIOstar® Plus microplate reader to carry out a high-throughput antiviral screen based on the inhibition of a viral cytopathic effect using commercially available cell-based assays such as the CellTiter-Glo® luminescent cell viability assay and CellTox™ Green fluorescent cell cytotoxicity assay. Both CellTiter-Glo® and CellTox™ Green assays provide a very robust, easy-to-use assay platform for the high-throughput screening of viral cytopathic effects. Thereby, molecules with antiviral activity against viruses such as SARS-CoV-2 and RSV that cause a viral cytopathic effect in infected cells can be identified.
Assay Principle
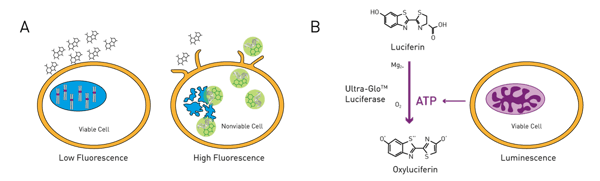
The CellTox™ Green cytotoxicity assay utilizes a cell membrane-impermeable dye, which is only able to enter non-viable cells where it binds to DNA and becomes fluorescent. This assay is not activity-based; the fluorescent signal is proportional to the number of dead cells in culture and thereby to the viral cytopathic effect. CellTiter-Glo® is a viability assay based on the quantification of ATP. Metabolically active cells provide ATP which is used to convert Luciferin to Oxyluciferin and light by the Ultra-Glo® Luciferase. Viral cytopathic effects can be assessed by the reduction of metabolic activity and decrease of the luminescence signal.
Materials & Methods
- NuncTM MicroWell™ 384 well, black, clear-bottom (Thermo Fisher, # 142761), for FI
- 384 well, white, clear bottom, PS treated plates (Corning®#CLS3765), for Lumi
- Echo® 525 liquid handler (Labcyte)
- Vero-E6 cells
- RSV virus stock (2x107/mL)
- SARS-CoV-2 virus stock (1.5x107/mL)
- CellTiter-Glo® luminescent cell viability assay kit (Promega #G7570)
- CellTox™ Green fluorescent cell cytotoxicity assay (Promega # G8741)
- CLARIOstar Plus microplate reader (BMG LABTECH)
Experimental Procedure
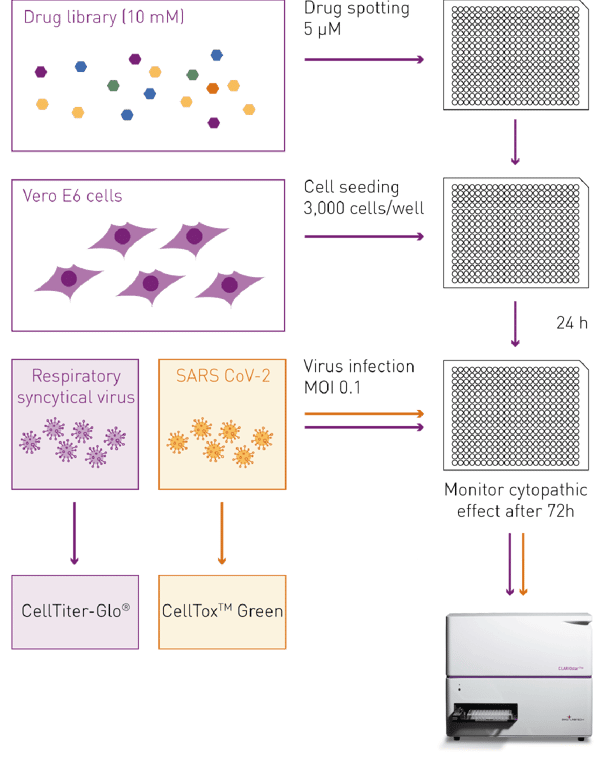
For the viral cytopathic effect assay, 25 nL of each drug was spotted into 384 well plates using the Echo liquid handler, and 50 µl of Vero-E6 cell suspension (3,000 cells per well) were added to obtain a final drug concentration of 5 µM. DMSO was used as a control. Following a 24 h incubation, the cells were infected with 5 µL of RSV or SARS-CoV-2 virus dilution which equals a multiplicity of infection (MOI) of 0.1. Infected cells were taken out of the incubator at 72 h post-infection and were analyzed for viral cytopathic effects by measuring cell cytotoxicity using CellTox™ Green or cell viability using CellTiter-Glo® on the CLARIOstar Plus plate reader.
Instrument settings
| CellTox™ Green fluorescent cell cytotoxicity assay | ||
| Optic settings | Fluorescence, endpoint, bottom optic | |
| General settings |
Monochromator settings | Excitation 483-14 Dichroic 502.5 Emission 530-30 |
| Gain | EDR | |
| Focal height | 2.5 mm | |
| Number of flashes per well | 20 | |
| Settling time (S) | 0.1 | |
| CellTiter-Glo® luminescent viability assay | ||
| Optic settings | Luminescence, endpoint, top optic | |
| General settings |
Filters | No filter |
| Gain | EDR | |
| Focal height | 13.5 mm | |
| Interval time (S) | 0.25, normalized to 1 | |
|
Aperture spoon |
96/384 | |
Results & Discussion
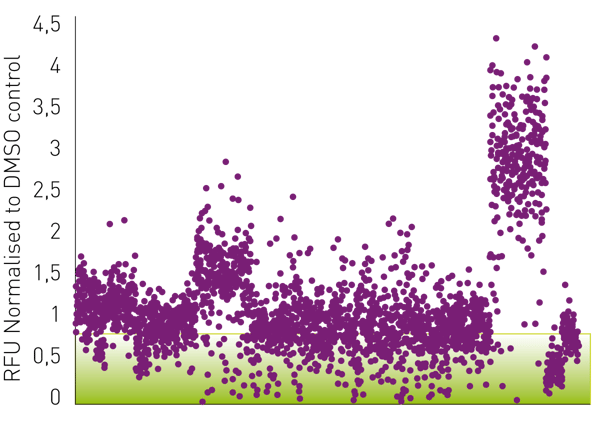
The viral cytopathic effect of SARS-CoV-2 and RSV on Vero-E6 cell cultures was assessed with a CellToxTM Green and a CellTiter-Glo® assay, respectively, 72 h post-infection. Fig. 3 shows the obtained data from the CellToxTM green assay performed on Vero-E6 cells infected with SARS-CoV-2. Drugs were classified as potential hits if they reduced the viral cytopathic effect below 90% (see green box) compared to the control samples, which were treated with DMSO and set to 1.
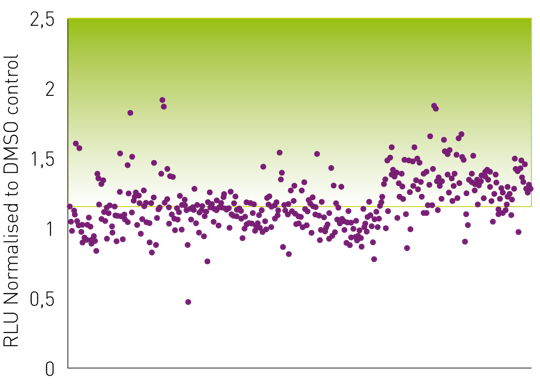
The graph below (fig. 4) shows the results of the CellTiter-Glo® assay. In contrast to the CellToxTM Green assay, the luminescence signal represents cell viability and thus virucidal drugs are expected to lead to an increase in signal. Here, the viability of above 1.1 (=110%) in comparison to the DMSO control (set to 1) was defined as a threshold to identify potential drugs inhibiting viral cytopathic effect.
Conclusion
The use of cytotoxicity/viability assays to study the impact of potential virucidal drugs on viral cytopathic effects offers several advantages. First, wild-type viruses can be applied since the evaluation takes place indirectly through the assessment of viral cytopathic effect, thereby eliminating the need for additional labelling or modification steps. Furthermore, running and evaluating luminescent and fluorescent assays on microplate readers like the CLARIOstar Plus, enables large libraries to be screened in high throughput and in far less time compared to traditional applications.
Microplate readers offer the ideal measurement platform for the identification of drugs, which effectively inhibit viral cytopathic effects allowing for the rapid elimination of 1000s of compounds that show no specific inhibition.
This approach has been used successfully by the University of Belfast to screen existing drugs and drug combinations.
References
- Touret, F. et al. In vitro screening of a FDA approved chemical library reveals potential inhibitors of SARS-CoV-2 replication. Sci Rep. doi: 10.1038/s41598-020-70143-6 (2020).
- Baer, A and Kehn-Hall, K. Viral concentration determination through plaque assays: using traditional and novel overlay systems. doi:10.3791/52065 (2014).