Introduction
The integrin family of proteins are essential for the ability of cells to adhere and to migrate on extracellular matrix components. There are two distinctive groups of integrins within the family that can be identified by their ability to bind to RGD containing extracellular matrix components such as fibronectin and those that act as collagen receptors. The migration of epithelial cell sheets can play an important role during development or wound healing. Phosphoinositides such as PtdIns3,4,5 P3 and PtdIns4,5P2 are thought to play a role in migration. To study migration in cell lines, scratch assays were typically performed where a confluent monolayer was scratched to remove a strip of cells and then the movement of the cells from the edges into the gap was quantified. This method has several drawbacks including the removal of the ECM components and the rupturing of cells which could all skew the results.
The OrisTM Cell Migration Assays specifically the TriCoated kits present certain advantages over the traditional migration assay including the ability to test the effect of different ECM components on migration. With the use of siRNA and the OrisTM TriCoated Cell Migration Assay we show here that phosphoinositides are important for migration. We were able to measure migration on different ECM components in real-time using a BMG LABTECH microplate reader fitted with a CO2 delivery system making it possible to keep cells in the plate reader for up to 48 hours.
Technology Overview
The OrisTM Cell Migration Assay is designed with unique OrisTM Cell Seeding Stoppers, Detection Mask, and Removal Tool. Simply seed the cells in each well, allow them to adhere, remove the Stoppers to create a detection zone, and measure the migration. The OrisTM Cell Migration Assay - TriCoated offers three surfaces (32 wells ach of Tissue Culture Treated, Collagen I, and Fibronectin) on a single 96-well microplate. More detailed information can be found on the Platypus website: www.platypustech.com/.
Materials & Methods
- DiI-C16 (Molecular Probes®, Invitrogen)
- siRNA (Dharmacon)
- OrisTM Cell Migration Assay (Platypus Technologies, LLC)
- Microplate reader (BMG LABTECH)
Cells were grown in DMEM obtained from Sigma with FCS, pen/strep, and glutamate from Invitrogen. Phenol red-free DMEM was obtained from Gibco. The osteosarcoma cell line U2OS was used in these assays.
Migration assay
U2OS cells were seeded in 6 well plates at 100000 cells/well and transfected the next day with 10 μL 20 μM siRNA per well along with 3 μL DharmaFECT. Negative control siRNA was also transfected separately. After overnight incubation, cells were trypsinised, washed, and counted. The total amount of cells needed for each assay was suspended in 1 mL phenol red-free medium containing 2.5 μM DiI and cells were incubated at 37°C for 30 minutes. Afterwards, cells were centrifuged and resuspended in phenol red-free medium to a concentration of 300000 cells/mL. Gel loading tips were used to deliver 100 μL of all suspension into each well of an OrisTM Cell Migration Assay TriCoated plate. For each condition, one well without a Cell Seeding Stopper served as the positive control. The plate was then incubated overnight at 37°C. The following day the Cell Seeding Stoppers were removed from test wells and the medium was flicked out. Subsequently, 200 μL phenol red-free medium was added to each well. The Detection Mask was attached to the bottom of the plate and the plate was put in the pre-warmed plate reader.
For all experiments the fluorescence was measured with excitation at 544 nm and emission at 590 nm. Readings were taken at 20-minute intervals over a period of 48 hours using the bottom optic. The gain was adjusted as 70 % of the positive control.
After completion of the assay, the data was analysed using the BMG LABTECH supplied MARS Data Analysis Software. First, a baseline correction was performed on the raw data using the data from the 6th to the 9th datapoints. Next, the curve was smoothed using moving averaging over three datapoints.
Results & Discussion
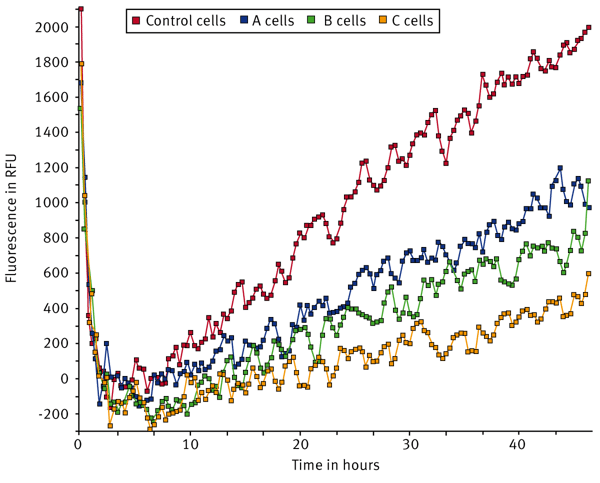
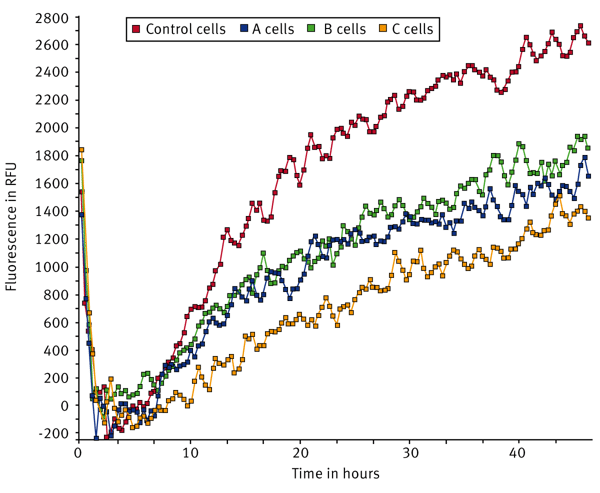
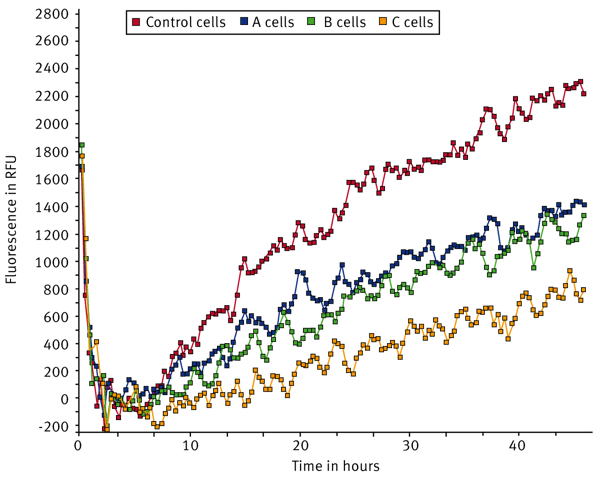
U2OS cells were transfected with a control non-targeting siRNA or with three different siRNA pools targeting specific proteins (A, B, C). Signal curves for tissue culture (TC) treated, Collagen I, and Fibronectin coated wells are shown in Fig. 2, 3, and 4.
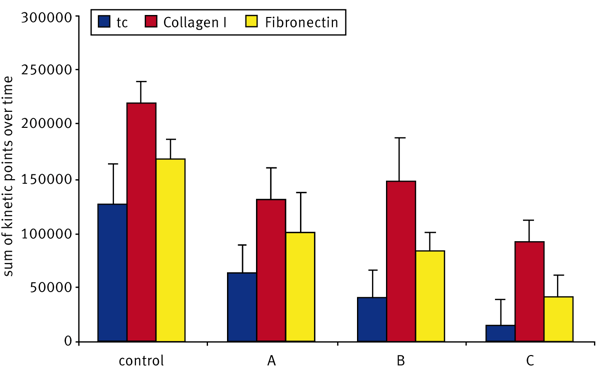
The sum function in the kinetic calculation window was used to determine the area under the curve for each sample. The sum values were averaged and the population standard deviation was calculated (Fig. 5).The data indicates that U2OS cells transfected with control siRNA migrate the fastest on Collagen I while migration was somewhat reduced in both fibronectin and TC-treated wells. Migration dropped by 34% in cells transfected with siRNA pool A on TC-treated wells while the drop is 41% on Collagen I and 45% on Fibronectin compared to that of the Control conditions. Cells transfected with the siRNA B pool demonstrated a similar drop in migration to siRNA pool A across all surfaces tested. The knockdown of siRNA pool C had a more pronounced effect with migration dropping 75% on TC-treated wells, 53% on Collagen I, and 69% on Fibronectin surfaces relative to the control. From this data, it is clear that the C siRNA construct inhibits migration to a large extent in cells plated on TC-treated wells and on Fibronectin while there was a somewhat smaller effect in cells on Collagen I.
Conclusion
This assay shows that the knockdown of genes A, B, or C has an effect on cell migration. Migration is inhibited on Tissue Culture Treated, Fibronectin, and Collagen I surfaces suggesting that the effect of these genes is not integrin-specific. Since all three proteins knocked down in these cells are involved in phosphoinositide metabolism we can conclude that phosphoinositides are of importance for cell migration.